Many regions in Qld and northern NSW are still experiencing an outbreak of Helicoverpa armigera in wheat.

Helicoverpa larvae in heads (left) and damage to grain (right)
Before the last significant outbreak occurred in 2016, it had been many years since anyone had seen such an event, and while there was a smaller outbreak in 2020, it was not on the scale or severity of this year. In many crops there are mixed infestation of Helicoverpa and native armyworm, which has raised questions about how to manage these pests, both individually, and in combination.
In 2016, the outbreak progressed south as temperatures warmed up enough for Helicoverpa to be active in crops.
Beatsheet articles on previous outbreaks:
- Helicoverpa in wheat – does it warrant control? (2020)
- High Helicoverpa armigera (corn earworm) activity now being seen in winter cereals (2016)
Insecticide registrations and resistance
It is important to note that the populations in wheat and barley crops (and in sorghum) are comprised entirely of H. armigera. This means that these populations will have moderate levels of resistance to carbamates (e.g. methomyl) and be highly resistant to synthetic pyrethroids (e.g. alpha cypermethrin).
Only methomyl and NPV (VivusMax®) are registered for helicoverpa control in winter cereals. Importantly, VivusMax® must be applied to larvae no larger than 10 mm in length (3rd instar).
Be aware of the withholding periods, which may differ depending on the markets into which grain will be sold. If in doubt, check with grain traders.
Read more about NSW DPI’s Helicoverpa resistance surveillance program.
Helicoverpa thresholds for winter cereals
When the 2016 outbreak occurred, there were no established thresholds because there had been no opportunities to do trial work previously. The DAF Field Crop Entomology team managed to undertake one replicated field trial to determine the damage potential of helicoverpa in wheat. The trial was established at St George under very high pressure, and subsequently experienced very high levels of crop loss where the infestation was not controlled.
The damage incurred included significant ‘defoliation’ (scarification) of leaves by smaller larvae in the canopy, resulting in reduced leaf function. Larger larvae (medium and bigger) moved into the heads and fed directly on grain. Trial work also demonstrated that larvae will continue to feed on grain even after physiological maturity, which is what happens in sorghum.
In combination, this damage resulted in the highest estimated crop loss that we have seen for helicoverpa – a whopping 42 kg/ha yield loss for each larva per square metre. In comparison, the yield loss in sorghum, chickpea and canola is estimated to be 24 kg/ha (direct grain feeding impacts) and a higher 36 kg/ha in mungbeans (a combination of bud and flower loss + direct grain feeding).
It would be of benefit to revisit this work to determine the relative contribution of the defoliation and direct grain feeding to overall crop loss. In 2022, infestations have mostly established in crops that are already at grain filling – so defoliation is not an issue in terms of contribution to loss. Consequently, rather than using the 42 kg/ha loss figure generated from impacts to both foliage and grain, it is more appropriate to use a loss figure that accounts only for losses directly to grain as it is filling and maturing. Because we this trial work has not been done in wheat/barley, we can only extrapolate from what we know about the losses that occur in other grain crops where direct grain feeding is the main cause of crop loss. The estimate of 24 kg/ha/larva per sqm is the figure that is proposed.
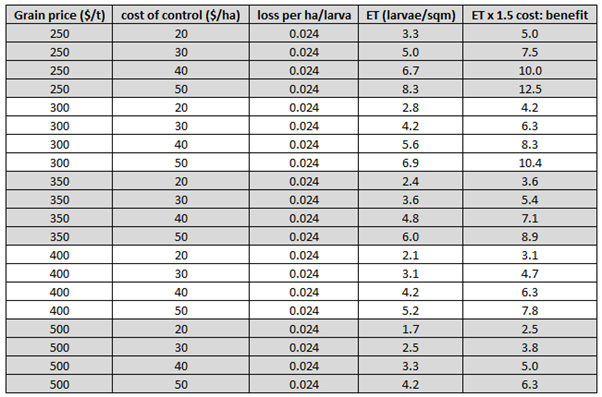
Table 1 shows the break-even threshold (cost=benefit) for helicoverpa in wheat/barley using a range of grain values and costs of control, along with an adjusted threshold for a better cost:benefit ratio.
Table 1. Ready reckoner for Helicoverpa thresholds in winter cereals using a range of grain values and costs of control.
If you would like to use different cost of control or grain value to calculate thresholds, the equation used and an example are provided below.
Additional factors to consider in prioritising at-risk crops:
- The higher yielding a crop, the more there is to protect
- The later a crop is, the longer it is susceptible to damage and loss.
- The longer a crop is green (leaf and head), the more likely small larvae are to successfully develop to damaging large larvae.
- The smaller the larvae, the more likely they are to die from predation, disease and impact of abiotic factors such as wind or rain (for example, the helicoverpa threshold calculator for chickpeas applies a 70% mortality/death factor to all VS-S larvae).
- The DARABUG development model is useful for estimating how much longer larvae of different ages will take to complete development.


Dipel DF is also registered for use in all crops for all lepidoptera species.
More suited to horticultural use than broadacre..
Dipel SC is not registered for winter cereals